Weil’s syndrome: from various organic dysfunctions to a favorable outcome
DOI:
https://doi.org/10.5935/2764-734X.e20240438Keywords:
Leptospirosis, Weil Disease, Acute Kidney Injury, Renal Dialysis, Case reportAbstract
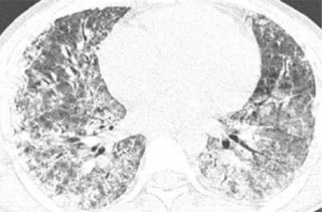
Human leptospirosis is the most widely distributed zoonosis globally. In tropical countries, it is an endemic disease, and outbreaks coincide with rainy periods, especially in flooded areas. In its early stages, early recognition of leptospirosis is challenged by the nonspecific signs and symptoms, making it necessary to value related epidemiological data to consider this diagnostic hypothesis, and subsequently, prescribe effective antimicrobial treatment from the beginning. Evolution of leptospirosis to a second stage with pulmonary, hepatic, and renal dysfunction determines a more serious and less frequent form of the disease, known as Weil’s syndrome. Herein, we describe a classic case of Weil’s syndrome, whose early recognition and appropriate treatment led to a satisfactory clinical response and total recovery, despite the initial severity of the condition.
Downloads
References
1. Gilden D, Cohrs RJ, Mahalingam R, Nagel MA. Varicella zoster virus vasculopathies: diverse clinical manifestations, laboratory features, pathogenesis, and treatment. Lancet Neurol. 2009;8(8):731-40. DOI: 10.1016/S1474-4422(09)70134-6
2. Marra CM. Infectious and postinfectious vasculopathies. Neuroimaging Clin N Am. 2024;34(1):13-21. DOI: 10.1016/j.nic.2023.06.001
3. Maher MD, Douglas VP, Douglas KAA, Collens SI, Gilbert AL, Torun N, et al. Clinical and neuroradiologic characteristics in varicella zoster virus reactivation with central nervous system involvement. J Neurol Sci. 2022;437:120262. DOI: 10.1016/j.jns.2022.120262
4. Gilden D, Nagel M, Cohrs R, Mahalingam R, Baird N. Varicella zoster virus in the nervous system. F1000Res. 2015;4(Faculty Rev-1356):1356. DOI: 10.12688/f1000research.7153.1
5. Grahn A, Studahl M. Varicella-zoster virus infections of the central nervous system – Prognosis, diagnostics and treatment. J Infect. 2015;71(3):281-93. DOI: 10.1016/j.jinf.2015.06.004
6. Nagel MA, Gilden D. Neurological complications of varicella zoster virus reactivation. Curr Opin Neurol. 2014;27(3):356-60. DOI: 10.1097/WCO.0000000000000092
7. Bakradze E, Kirchoff KF, Antoniello D, Springer MV, Mabie PC, Esenwa CC, et al. Varicella zoster virus vasculitis and adult cerebrovascular disease. Neurohospitalist. 2019;9(4):203-208. DOI: 10.1177/1941874419845732
8. Nagel MA, Traktinskiy I, Azarkh Y, Kleinschmidt-DeMasters B, Hedley-Whyte T, Russman A et al. Varicella zoster virus vasculopathy: analysis of virus-infected arteries. Neurology. 2011;77(4):364-70. DOI: 10.1212/WNL.0b013e3182267bfa
9. Gilden D. Varicella-zoster virus infections. Continuum (Minneap Minn). 2015;21(6):1692-703. DOI: 10.1212/CON.0000000000000246
10. Carod Artal FJ. Clinical management of infectious cerebral vasculitides. Expert Rev Neurother. 2016;16(2):205-21. DOI: 10.1586/14737175.2015.1134321
11. Kennedy PG, Mogensen TH. Determinants of neurological syndromes caused by varicella zoster virus (VZV). J Neurovirol. 2020;26(4):482-95. DOI: 10.1007/s13365-020-00857-w
12. Chiang F, Panyaping T, Tedesqui G, Sossa D, Costa Leite C, Castillo M. Varicella zoster CNS vascular complications. A report of four cases and literature review. Neuroradiol J. 2014;27(3):327-33. DOI: 10.15274/NRJ-2014-10037
13. Nagel MA, Niemeyer CS, Bubak AN. Central nervous system infections produced by varicella zoster virus. Curr Opin Infect Dis. 2020;33(3):273-8. DOI: 10.1097/QCO.0000000000000647
14. Cheng-Ching E, Jones S, Hui FK, Man S, Gilden D, Bhimraj A, et al. High-resolution MRI vessel wall imaging in varicella zoster virus vasculopathy. J Neurol Sci. 2015;351(1-2):168-73. DOI: 10.1016/j.jns.2015.02.017
15. Langan SM, Minassian C, Smeeth L, Thomas SL. Risk of stroke following herpes zoster: a self-controlled case-series study. Clin Infect Dis. 2014;58(11):1497-503. DOI: 10.1093/cid/ciu098
16. Nagel MA, Gilden D. Developments in varicella zoster virus vasculopathy. Curr Neurol Neurosci Rep. 2016;16(2):12. DOI: 10.1007/s11910-015-0614-5
17. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde e Ambiente. Departamento de Imunizações e Doenças Imunopreveníveis. Manual dos Centros de Referência para Imunobiológicos Especiais [Internet]. 6ª ed. Brasília: Ministério da Saúde; 2023; [acesso em 31 de janeiro de 2024]. Disponível em: https://sbim.org.br/images/calendarios/manual-dos-centros-de-referencia-para-imunobiologicos-especiais-6a-edicao-2023.pdf
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Infections in Evidence

This work is licensed under a Creative Commons Attribution 4.0 International License.
All users may freely read, download, share, and adapt this scientific product for any purpose (even commercial ones), provided that due credit is given to the authors and the original publication, and that any alterations are duly indicated.
> Ethics
All articles published herein have received ethical approval from the National Research Ethics System (formerly CEP/CONEP system) based on Federal Law 14.874/24 and other specific Brazilian regulations, or a similar document attesting to the knowledge and authorization from the institution of origin in the case of foreign works.
The authors declare that there is no sponsorship or conflict of interest of any kind, unless otherwise indicated in the body of the article.
It is worth noting that case reports are a valuable learning resource for the scientific community, but should not be used in isolation to guide diagnostic or therapeutic options in clinical practice or health policies
















