Mycobacterium abscessus infection after aesthetic surgical procedure
DOI:
https://doi.org/10.5935/2764-734X.e20231030Keywords:
Mycobacterium abscessus, Mycobacterium Infections, Nontuberculous, Postoperative Complications, Plastic Surgery Procedures, Case ReportAbstract
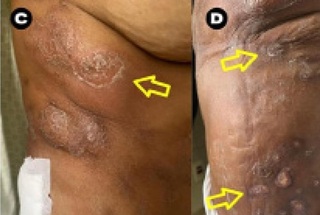
Infections caused by Mycobacterium abscessus are uncommon, however, cases are emerging, especially associated with trauma and infections related to health care after aesthetic surgical procedures. The diagnosis is difficult and the treatment is long time. Although mortality rates are low, it is associated with significant morbidity, The authors describe a case of Mycobacterium abscessus infection with myocutaneous manifestation, after abdominal liposuction and gluteal graft with diffuse ulcer-crusted lesions, subcutaneous and myofascial collections, fasciitis and constitutional symptoms. The diagnosis was obtained by culture of secretion. The treatment was started with amikacin, tigecycline, clarithromycin and ertapenem and the maintenance with amikacin, clarithromycin and clofazimine, with regresson of the lesions.
Downloads
References
1. Lyon S, Moura ACL, Grossi MAF, Silva, RCV. Micobacterioses. In: Dermatologia tropical. 1ª ed. Rio de Janeiro: Medbook Editora Científica; 2017. p. 38-41.
2. Nogueira LB, Garcia CN, Costa MSCD, Moraes MB, Kurizky PS, Gomes CM. Non-tuberculous cutaneous mycobacterioses. An Bras Dermatol. 2021;96(5):527-38.
3. Ministério da Saúde (BR). Agência Nacional de Vigilância Sanitária (Anvisa). Boletim Segurança do Paciente e Qualidade em Serviço de Saúde no 19 GVIMS/GGTES/Anvisa: Notificação de casos de micobactéria de crescimento rápido (MCR) atualizado - 1998 a 25 de julho de 2023. Brasília (DF): Ministério da Saúde; 2023; [acesso em 2023 Abr 02]. Disponível em: https://app.powerbi.com/view?r=eyJrIjoiYWU2MTQ3OWItZjAwMy00ZTNkLTk2NzAtMzExMjM0ODc5M2Y0IiwidCI6ImI2N2FmMjNmLWMzZjMtNGQzNS04MGM3LWI3MDg1ZjVlZGQ4MSJ9
4. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de Doenças de Condições Crônicas e Infecções Sexualmente Transmissíveis. Recomendações para o diagnóstico e tratamento das doenças causadas por micobactérias não tuberculosas no Brasil. Brasília (DF): Ministério da Saúde; 2021; [acesso em 2023 Abr 02]. Disponível em: https://www.gov.br/aids/pt-br/central-de-conteudo/publicacoes/2021/recomendacoes-para-o-diagnostico-e-tratamento-das-doencas-causadas-por-micobacterias-nao-tuberculosas-no-brasil
5. Sousa PP, Cruz RCS, Schettini APM, Westpha DC. Mycobacterium abscessus skin infection after tattooing - case report. An Bras Dermatol. 2015;90:739-41.
6. Ryu HJ, Kim WJ, Oh CH, Song HJ. Iatrogenic Mycobacterium abscessus infection associated with acupuncture: clinical manifestations and its treatment. Int J Dermatol. 2005;44:846-50.
7. Fitzgerald DA, Smith AG, Lees A, Yee L, Cooper N, Harris SC, et al. Cutaneous infection with Mycobacterium abscessus. Br J Dermatol. 1995;132(5):800-4.
8. Fang RY, Sun QN. Mycobacterium abscessus infections following injection of botulinum toxin. J Cosmet Dermatol. 2020;19(4):817-9.
9. Johansen MD, Herrmann JL, Kremer L. Non-tuberculous mycobacteria and the rise of Mycobacterium abscessus. Nat Rev Microbiol. 2020;18:392-407.
10. Boudehen YM, Kremer L. Mycobacterium abscessus. Trends Microbiol. 2021;29:951-2.
11. Victoria L, Gupta A, Gómez JL, Robledo J. Mycobacterium abscessus complex: a review of recent developments in an emerging pathogen. Front Cell Infect Microbiol. 2021;11:659997.
12. Carvalho NFG, Ferrazoli L, Riveron MBA, Chimara E. Caracterização dos surtos causados pelo grupo Mycobacterium abscessus. Rev Inst Adolfo Lutz. 2012;71(2):228-36.
13. Fröberg G, Maurer FP, Chryssanthou E, Fernström L, Benmansour H, Boarbi S, et al; EUCAST AMST and ESCMYC study groups. Towards clinical breakpoints for non-tuberculous mycobacteria - Determination of epidemiological cut off values for the Mycobacterium avium complex and Mycobacterium abscessus using broth microdilution. Clin Microbiol Infect. 2023 Jun;29(6):758-64.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Infections in Evidence

This work is licensed under a Creative Commons Attribution 4.0 International License.
All users may freely read, download, share, and adapt this scientific product for any purpose (even commercial ones), provided that due credit is given to the authors and the original publication, and that any alterations are duly indicated.
> Ethics
All articles published herein have received ethical approval from the National Research Ethics System (formerly CEP/CONEP system) based on Federal Law 14.874/24 and other specific Brazilian regulations, or a similar document attesting to the knowledge and authorization from the institution of origin in the case of foreign works.
The authors declare that there is no sponsorship or conflict of interest of any kind, unless otherwise indicated in the body of the article.
It is worth noting that case reports are a valuable learning resource for the scientific community, but should not be used in isolation to guide diagnostic or therapeutic options in clinical practice or health policies
















