Atypical presentation of cutaneous leishmaniasis in a pregnant woman
DOI:
https://doi.org/10.5935/2764-734X.e20240541Keywords:
Cutaneous leishmaniasis, Amphotericin B, Meglumine antimoniate, Pregnancy complications, Infections, Case reportAbstract
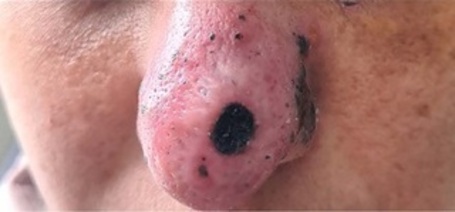
Cutaneous leishmaniasis most commonly presents as a single cutaneous ulcer, but there are less common clinical presentations. Here we report a rare manifestation of cutaneous leishmaniasis diagnosed in a 37-year-old pregnant woman who presented with an infiltrative, erythematous, raspberry-like lesion on the nasal dorsum of three months’ duration. No mucosal involvement was noted, but the patient complained of local pain and paresthesias. There was no history of immunosuppression or other comorbidities. Initially treated with amphotericin B, the patient experienced a relapse after eight months, at which time we decided to restart treatment with pentavalent antimonial. The delay in diagnosis caused by the atypical presentation of the disease led to a delay in treatment. We emphasize the importance of including cutaneous leishmaniasis in the differential diagnosis of any skin lesion in endemic areas.
Downloads
References
1. Ministério da Saúde (BR). Secretaria de Vigilância em Saúde. Departamento de Vigilância das Doenças Transmissíveis. Manual de Vigilância da Leishmaniose Tegumentar Americana. 1a ed. Brasília: Ministério da Saúde; 2017 [acesso em 2024 Fev 26]. Disponível em: https://bvsms.saude.gov.br/bvs/publicacoes/manual_vigilancia_leishmaniose_tegumentar.pdf
2. Goto H, Lindoso JA. Current diagnosis and treatment of cutaneous and mucocutaneous leishmaniasis. Expert Rev Anti Infect Ther. 2010;8(4):419-33. DOI: 10.1586/eri.10.19
3. Marchi MNA de, Caldart ET, Martins FDC, Freire RL. Spatial analysis of leishmaniasis in Brazil: a systematized review. Rev Inst Med trop S Paulo. 2019;61:e68. DOI: 10.1590/S1678-9946201961068
4. Meireles CB, Maia LC, Soares GC, Teodoro IPP, Gadelha MDSV, da Silva CGL, et al. Atypical presentations of cutaneous leishmaniasis: a systematic review. Acta Trop. 2017:172:240-54. DOI: 10.1016/j.actatropica.2017.05.022
5. van Griensven J, Carrillo E, López-Vélez R, Lynen L, Moreno J. Leishmaniasis in immunosuppressed individuals. Clin Microbiol Infect. 2014;20(4):286-99. DOI: 10.1111/1469-0691.12556
6. Von Stebut E. Immunology of cutaneous leishmaniasis: the role of mast cells, phagocytes and dendritic cells for protective immunity. Eur J Dermatol. 2007;17(2):115-22. DOI: 10.1684/ejd.2007.0122
7. Aluvihare VR, Kallikourdis M, Betz AG. Regulatory T cells mediate maternal tolerance to the fetus. Nat Immunol. 2004;5(3):266-71. DOI: 10.1038/ni1037
8. Mor G, Cardenas I. The immune system in pregnancy: a unique complexity. Am J Reprod Immunol. 2010;63(6):425-33. DOI: 10.1111/j.1600-0897.2010.00836.x
9. Pokutnaya D, Shirzadi MR, Salari E, Molaei G. Cutaneous Leishmaniasis during Pregnancy, Preterm Birth, and Neonatal Death: A Case Report. Iran J Parasitol. 2020;15(4):608-14. DOI: 10.18502/ijpa.v15i4.4875
10. Morgan DJ, Guimaraes LH, Machado PR, D’Oliveira A Jr, Almeida RP, Lago EL, et al. Cutaneous leishmaniasis during pregnancy: exuberant lesions and potential fetal complications. Clin Infect Dis. 2007;45(4):478-82. DOI: 10.1086/520017
11. de Vries HJC, Schallig HD. Cutaneous Leishmaniasis: A 2022 Updated Narrative Review into Diagnosis and Management Developments. Am J Clin Dermatol. 2022;23(6):823-40. DOI: 10.1007/s40257-022-00726-8
12. Graça GC, Volpini AC, Romero GA, Oliveira Neto MP, Hueb M, Porrozzi R, et al. Development and validation of PCR-based assays for diagnosis of American cutaneous leishmaniasis and identification of the parasite species. Mem Inst Oswaldo Cruz. 2012;107(5):664-74. DOI: 10.1590/s0074-02762012000500014
13. Almeida OL, Santos JB. Advances in the treatment of cutaneous leishmaniasis in the new world in the last ten years: a systematic literature review. An Bras Dermatol. 2011;86(3):497-506. DOI: 10.1590/s0365-05962011000300012
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Infections in Evidence

This work is licensed under a Creative Commons Attribution 4.0 International License.
All users may freely read, download, share, and adapt this scientific product for any purpose (even commercial ones), provided that due credit is given to the authors and the original publication, and that any alterations are duly indicated.
> Ethics
All articles published herein have received ethical approval from the National Research Ethics System (formerly CEP/CONEP system) based on Federal Law 14.874/24 and other specific Brazilian regulations, or a similar document attesting to the knowledge and authorization from the institution of origin in the case of foreign works.
The authors declare that there is no sponsorship or conflict of interest of any kind, unless otherwise indicated in the body of the article.
It is worth noting that case reports are a valuable learning resource for the scientific community, but should not be used in isolation to guide diagnostic or therapeutic options in clinical practice or health policies
















